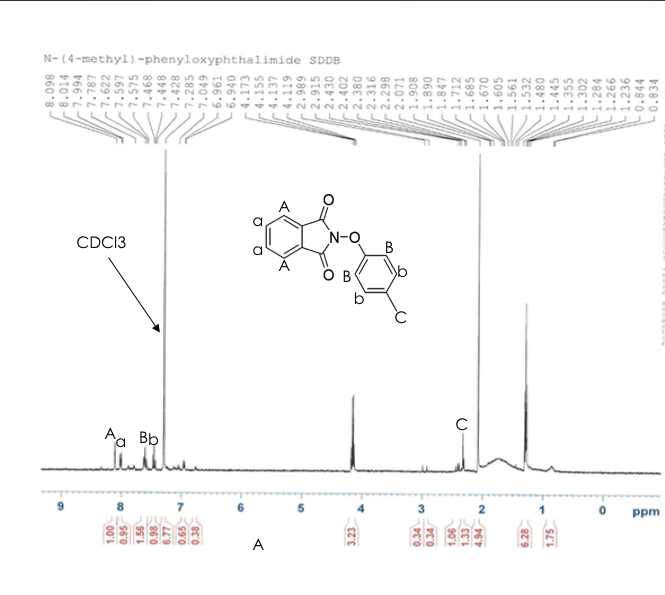
N-phenyloxyphthalimide, from step two, was successfully synthesized after the second trial. After the first attempt in the synthesis of N-phenyloxyphthalimide a H-NMR was taken in order to determine if the right compound was created. The H-NMR scan in figure 10 demonstrates that the compound is present but in very low yields. After the second trial, a H-NMR was once again conducted. This H-NMR had more distinctive peaks. The second trial also had a product yield at 80%. In order to take the H-NMR, chloroform-d was added into the mixture to prepare a H-NMR sample. The mixture of the chloroform-d and our compound was then tested in the H-NMR machine. The H-NMR results can be seen in figure _. The rotary evaporator was then used at a temperature around 60 °C in order to remove the chloroform. Once the chloroform was removed, the compound and the round bottom flask were measured and were about 74.48 grams. The compound was then scraped out of the flask, the flask was washed and weighed again after it was dried. The amount of compound was then found by subtracting the weight of the compound and flask from the weight of the flask. It was derived that 0.2057 grams of the N-phenyloxyphthalimide were created.
Although we got an 80% yield, after trial two, the H-NMR spectroscopy scan reveals that very little of N-phenyloxyphthalimide was actually created. The peaks on the H-NMR that were taken demonstrate that there are other compounds besides N-phenyloxyphthalimide in the mixture. This means that the compound that was desired from this step, N-phenyloxyphthalimide, was created but was impure . The H-NMR from trial two is shown below in figure (10).
Although we got an 80% yield, after trial two, the H-NMR spectroscopy scan reveals that very little of N-phenyloxyphthalimide was actually created. The peaks on the H-NMR that were taken demonstrate that there are other compounds besides N-phenyloxyphthalimide in the mixture. This means that the compound that was desired from this step, N-phenyloxyphthalimide, was created but was impure . The H-NMR from trial two is shown below in figure (10).
Figure 10. H-NMR after Step 2
In the H-NMR above, the peaks between 7.4 and 6.8 correlate with our compound but compared to the other peaks the peaks between 7.4 and 6.8 are significantly smaller revealing that very little of was synthesized in step two. Step three and four were not conducted because of time and material limitations.
Although we did not conduct step three and four, the data gathered from steps one and two prove that the synthesis of O-(4-methyl)-phenyl-N-(9’acridnyl)-hydroxylamine will be extremely difficult because of the unwanted compounds also included in the mixture. The low yields from step one and two could also indicate that a new process in step two is needed to get rid of the DMF and other impurities. In addition, step three and four will need to be tested in order to determine their efficiency. More research proves that there is more than one process to synthesize the compound needed for step three. The processes are further outlined in the DeSelm’s paper. Although the reaction was incomplete, it is predicted that the synthesis of the compound is possible under inert and specific conditions.
Although we did not conduct step three and four, the data gathered from steps one and two prove that the synthesis of O-(4-methyl)-phenyl-N-(9’acridnyl)-hydroxylamine will be extremely difficult because of the unwanted compounds also included in the mixture. The low yields from step one and two could also indicate that a new process in step two is needed to get rid of the DMF and other impurities. In addition, step three and four will need to be tested in order to determine their efficiency. More research proves that there is more than one process to synthesize the compound needed for step three. The processes are further outlined in the DeSelm’s paper. Although the reaction was incomplete, it is predicted that the synthesis of the compound is possible under inert and specific conditions.