Methods
The synthesis of O-(4-methyl)-phenyl-N-(9'-acridinyl)-hydroxylamine is a multiple step product that consists of four steps. The first step, Synthesis of Diaryliodonium Triflate, is synthesized from p-iodotoluene and toluene as demonstrated in figure (6). Next N-(4-methyl)-phenyloxyphthalimide is synthesized by diaryliodonium salt and N-hydroxyphthalmine as shown in figure(insert number here). The next step, shown in figure(insert number here), is the Hydrolysis of O-(4-methyl)-phenylhydroxylamine. The final step of the reaction is the synthesis of O-(4-methyl)-phenyl-N-(9'-acridinyl)-hydroxylamine is outlined in figure(insert number here). Most of the steps used in the methods are outlined in DeSelm’s paper as well as other sources (4).
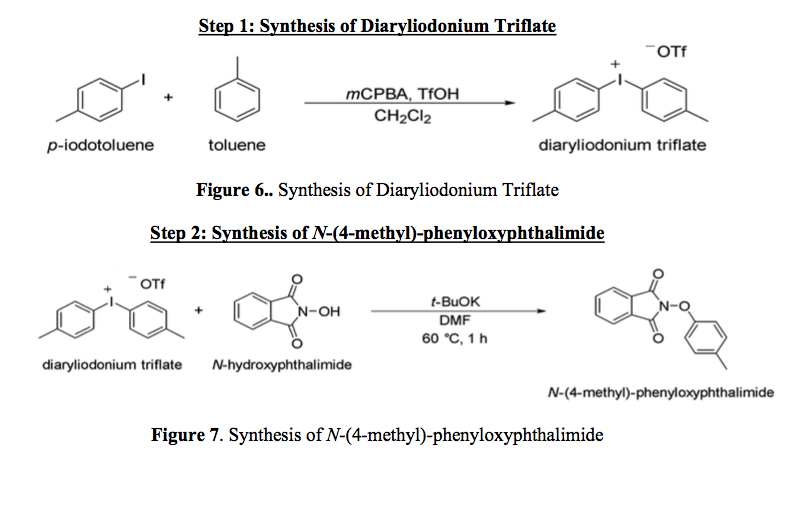
Synthesis of Diaryliodonium Triflate
This step is based on the procedure outlined by DeSelm as well as Bielwaski et al (2). The first step in the synthesis of Diaryliodonium Triflate will begin with a 25 mL round bottom flask which is stoppered. In order to perform the reaction under inert conditions, argon gas was inserted into the flask as well. Then 121 mg of m-chloroperbenzoic acid (mCPBA) and 2 mL of dichloromethane were added into the flask. Next 100.4 mg of p-iodotoluene to the flask and 0.047913 mL of toluene were added to the flask and the stir magnet was turned on. 120µL (3 equivalents) of trifluoromethanesulfonic acid (TfOH) was added into the flask via needle, after that the mixture turned dark green. Once TfOH was added into the flask, the mixture was left to stir for ten minutes. After the reaction was completed, the rotary evaporator was used to separate the solvent. Then, diethyl ether was measured to 2 mL and was added to the concentrated solution and stirred for ten minutes once again. The flask was then placed in the refrigerator overnight. This reaction is illustrated in figure 7.
This step is based on the procedure outlined by DeSelm as well as Bielwaski et al (2). The first step in the synthesis of Diaryliodonium Triflate will begin with a 25 mL round bottom flask which is stoppered. In order to perform the reaction under inert conditions, argon gas was inserted into the flask as well. Then 121 mg of m-chloroperbenzoic acid (mCPBA) and 2 mL of dichloromethane were added into the flask. Next 100.4 mg of p-iodotoluene to the flask and 0.047913 mL of toluene were added to the flask and the stir magnet was turned on. 120µL (3 equivalents) of trifluoromethanesulfonic acid (TfOH) was added into the flask via needle, after that the mixture turned dark green. Once TfOH was added into the flask, the mixture was left to stir for ten minutes. After the reaction was completed, the rotary evaporator was used to separate the solvent. Then, diethyl ether was measured to 2 mL and was added to the concentrated solution and stirred for ten minutes once again. The flask was then placed in the refrigerator overnight. This reaction is illustrated in figure 7.
Synthesis of N-(4-methyl)-phenyloxyphthalimide
The second step in the synthesis of O-(4-methyl)-phenyl-N-(9'-acridinyl)-hydroxylamine starts with a 25 mL round bottom flask inserted with argon gas in order to mimic inert atmosphere conditions. These steps are detailed by Ghosh et. al. 2 mL of anhydrous dimethylformamide (DMF) and 61.7 mg of Potassium t-butoxide (t-BuOK) were added to the flask (8). Next, 82 mg of Hydroxyphthalimide was dissolved in the round bottom flask. The flask was then left to stir for ten minutes so the reaction could occur. Then the diaryliodonium salt from step one was added into the mixture. In order to get all the diaryliodonium salt out, the flask was rinsed with DMF. The solution was then heated to 60 °C and left to heat for one hour. After an hour the mixture was cooled to room temperature and added into a separatory funnel in order to separate the aqueous layer from the organic layer. 20 mL of deionized water and 20 mL of ethyl acetate were added to the separatory funnel to aid in the separation of the two layer. Once the two liquids were added, the separatory funnel was shaken then left to separate into two layers. Once the two layers were split, the aqueous layer was removed. 20 mL of water was once again added and shaken inside of the separatory funnel. After draining the aqueous layer once again. The solution was once again washed with 20 mL of concentrated brine solution in order to separate the rest of the aqueous layer from the organic layer. Once the aqueous layer is drained, the organic layer was poured into an Erlenmeyer flask and was dried using anhydrous sodium sulfate. Once the anhydrous sodium sulfate could not retain any more water, it was filtered out using a gravity filtration system. In order remove the solution stuck in the anhydrous sulfate, ethyl acetate can be rinsed through the anhydrous sulfate. Ethyl acetate can be used in order to get the solution stuck in the anhydrous sulfate out. Next, the mixture is placed in the rotary evaporator in order to remove the ethyl acetate. After this step, there is an orange powder in the flask. In order to make sure the compound was formed an NMR was taken and can be seen in Figure 8.
The second step in the synthesis of O-(4-methyl)-phenyl-N-(9'-acridinyl)-hydroxylamine starts with a 25 mL round bottom flask inserted with argon gas in order to mimic inert atmosphere conditions. These steps are detailed by Ghosh et. al. 2 mL of anhydrous dimethylformamide (DMF) and 61.7 mg of Potassium t-butoxide (t-BuOK) were added to the flask (8). Next, 82 mg of Hydroxyphthalimide was dissolved in the round bottom flask. The flask was then left to stir for ten minutes so the reaction could occur. Then the diaryliodonium salt from step one was added into the mixture. In order to get all the diaryliodonium salt out, the flask was rinsed with DMF. The solution was then heated to 60 °C and left to heat for one hour. After an hour the mixture was cooled to room temperature and added into a separatory funnel in order to separate the aqueous layer from the organic layer. 20 mL of deionized water and 20 mL of ethyl acetate were added to the separatory funnel to aid in the separation of the two layer. Once the two liquids were added, the separatory funnel was shaken then left to separate into two layers. Once the two layers were split, the aqueous layer was removed. 20 mL of water was once again added and shaken inside of the separatory funnel. After draining the aqueous layer once again. The solution was once again washed with 20 mL of concentrated brine solution in order to separate the rest of the aqueous layer from the organic layer. Once the aqueous layer is drained, the organic layer was poured into an Erlenmeyer flask and was dried using anhydrous sodium sulfate. Once the anhydrous sodium sulfate could not retain any more water, it was filtered out using a gravity filtration system. In order remove the solution stuck in the anhydrous sulfate, ethyl acetate can be rinsed through the anhydrous sulfate. Ethyl acetate can be used in order to get the solution stuck in the anhydrous sulfate out. Next, the mixture is placed in the rotary evaporator in order to remove the ethyl acetate. After this step, there is an orange powder in the flask. In order to make sure the compound was formed an NMR was taken and can be seen in Figure 8.
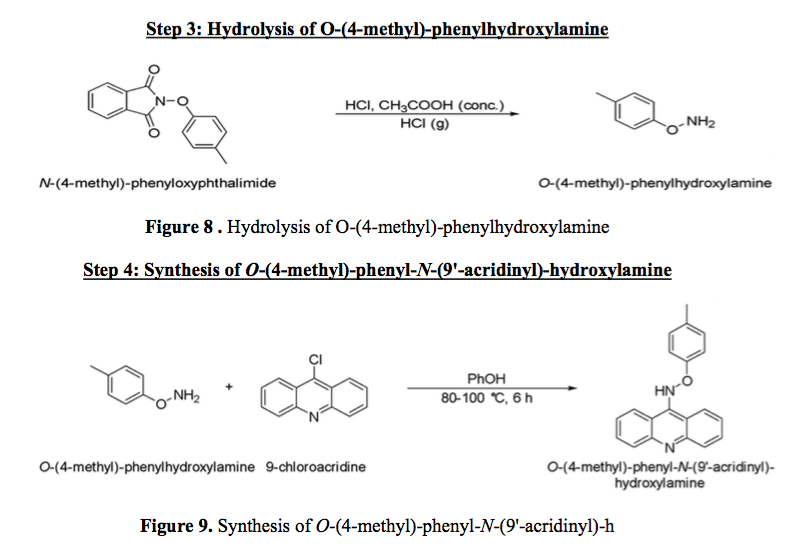
Hydrolysis of O-(4-methyl)-phenylhydroxylamine
In order to do step three shown in figure 8, N-(4-methyl)-phenyloxyphthalimide was added to concentrated hydrochloric acid (HCL, 12.1 M) and 15 mL of glacial acid. This process was done by the method outlined by Carlson (). The mixture was then allowed to reflux for two hours. In order to remove the rest of the solvent, the rotary evaporator is once again used. Next, the powder is scraped into a 50mL Erlenmeyer Flask. The residue in the flask is washed out using ionized water and poured into the Erlenmeyer flask. Then sodium hydroxide solution was added using a pipette in dropwise addition. Then the whole solution was poured into a separatory funnel, and 30 mL of dichloromethane and 20 mL of deionized water was used to separate the liquid into two layers. The solution was dried using anhydrous sodium sulfate once again and a gravity filter was used to separate the anhydrous sodium sulfate from the rest of the solution. Next HCl was bubbled through the mixture and left to stir for 30 minutes. Next, a suction filter was used to separate the solvent and the mixture was left to drive overnight.
In order to do step three shown in figure 8, N-(4-methyl)-phenyloxyphthalimide was added to concentrated hydrochloric acid (HCL, 12.1 M) and 15 mL of glacial acid. This process was done by the method outlined by Carlson (). The mixture was then allowed to reflux for two hours. In order to remove the rest of the solvent, the rotary evaporator is once again used. Next, the powder is scraped into a 50mL Erlenmeyer Flask. The residue in the flask is washed out using ionized water and poured into the Erlenmeyer flask. Then sodium hydroxide solution was added using a pipette in dropwise addition. Then the whole solution was poured into a separatory funnel, and 30 mL of dichloromethane and 20 mL of deionized water was used to separate the liquid into two layers. The solution was dried using anhydrous sodium sulfate once again and a gravity filter was used to separate the anhydrous sodium sulfate from the rest of the solution. Next HCl was bubbled through the mixture and left to stir for 30 minutes. Next, a suction filter was used to separate the solvent and the mixture was left to drive overnight.
Synthesis of O-(4-methyl)-phenyl-N-(9'-acridinyl)-hydroxylamine
The processes in this step are described by DeSelm (5). The final step of the reaction begins with 3.3 g of molten phenol (PhOH) and 9-chloroacridine was added to a round bottom flask with a stir bar. O-(4-methyl)-phenylhydroxylamine is also added into this flask. Then, this flask is then heated to 80-90 °C and left for six hours. After six hours the flask is cooled to room temperature and added to 50 mL of CH2Cl2. Next, the mixture and 100 mL of sodium hydroxide solution are added to the separatory funnel and the excess phenol is removed. Once the excess phenol is removed, the organic mixture will be dried using anhydrous MgSO4 and then gravity filtered to remove the anhydrous MgSO4. In order to purify the substance, the mixture will be placed in the rotary evaporator.
The processes in this step are described by DeSelm (5). The final step of the reaction begins with 3.3 g of molten phenol (PhOH) and 9-chloroacridine was added to a round bottom flask with a stir bar. O-(4-methyl)-phenylhydroxylamine is also added into this flask. Then, this flask is then heated to 80-90 °C and left for six hours. After six hours the flask is cooled to room temperature and added to 50 mL of CH2Cl2. Next, the mixture and 100 mL of sodium hydroxide solution are added to the separatory funnel and the excess phenol is removed. Once the excess phenol is removed, the organic mixture will be dried using anhydrous MgSO4 and then gravity filtered to remove the anhydrous MgSO4. In order to purify the substance, the mixture will be placed in the rotary evaporator.
Fusce in sapien scelerisque, mollis urna ut, vulputate sem. Nunc sollicitudin eget nisl maximus suscipit. Maecenas aliquam lectus sit amet justo lacinia rutrum. |